SUSTech scientists collaborate on new pancreatic cancer treatment targets
2019-04-18
Pancreatic cancer is thought to be the “king of cancers,” in that it has no obvious symptoms in its earliest stages, and is often only diagnosed after the tumor has metastasized and spread throughout the entire body. It often responds badly to chemotherapy as the tumor cells are usually located with the dense stroma, or structural support for the pancreas. The lack of effective targeted drugs and biomarkers also hampers treatment. This is mainly due to a unique microenvironment formed from dense interstitial cells, creating a protective barrier which gives the tumors significant resistance to most anti-cancer medications.
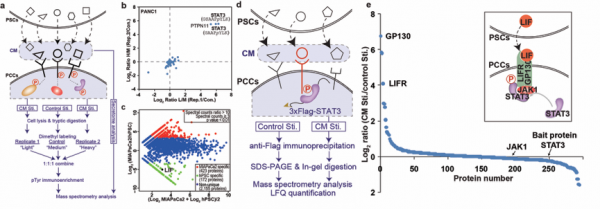
Associate Professor Tian Ruijun’s research group from the Department of Chemistry at Southern University of Science and Technology (SUSTech) joined a global effort over a long period of time to find new ways to target pancreatic cancer cells. Working with the Salk Institute for Biological Sciences, the collective group found that an interaction between pancreatic stellate cells (PSC) and pancreatic cancer cells (PCCs) could be taken advantage of through a key protein. This research was published today in Nature, under the title, “Targeting LIF-mediated paracrine interaction for pancreatic cancer therapy and monitoring.”
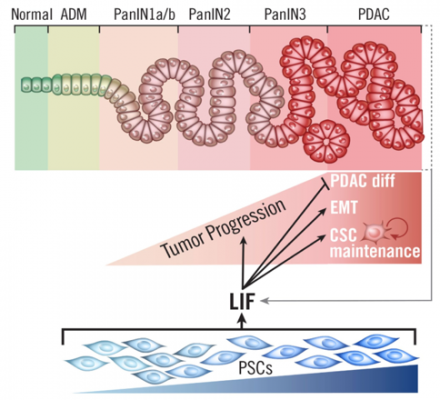
The PSC in the interstitial cell layer of PCCs can represent up to 90% of the mass of a pancreatic cancer tumor. The activation of PSC not only forms the protective barrier around the tumor, but also produces a specific signaling protein called Leukemia Inhibitory Factor (LIF). The LIF protein drives the development and progression of pancreatic cancer by stimulating cancer cells.
The researchers found that LIF protein levels are undetectable in a normal pancreas, but highly elevated in the tissue of patients with pancreatic ductal adenocarcinoma (PDAC), the most common form of pancreatic cancer. The tumor cells infect one or some of the ducts inside the pancreas and then spread from there. The elevated LIF levels in both mouse and human pancreatic cancer tissue indicated that activated PSC were responsible for producing LIF.
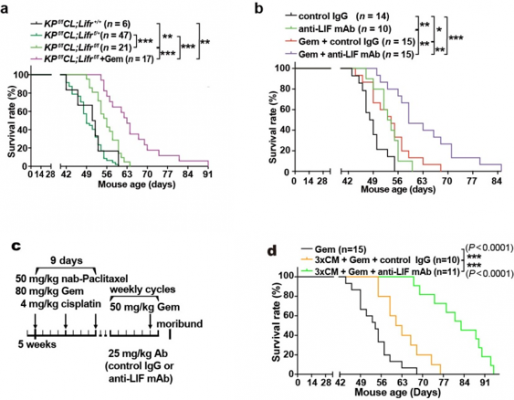
By selectively blocking the LIF receptor, the research teams were able to reduce the impact of PSC on PCCs, and in combination with other treatments, the survival time of mice was prolonged.
This research indicates that there are exciting opportunities for further research in this area. The effectiveness of targeting LIF, in conjunction with chemotherapy or other treatment methods, marks a new approach to pancreatic cancer. SUSTech developed an integrated proteomics analysis strategy that has provided unique system-level research tools for big data that allowed the research teams to better understand intercellular signaling within the tumor microenvironment. They believe that this cutting-edge achievement will help researchers pass the previous bottleneck associated with pancreatic cancer treatment and diagnosis.
The Food and Drug Administration (USA) has already approved a Phase I clinical trial by Northern Biologics Canada, based in part on the pivotal research conducted by SUSTech and the Salk Institute. They will test the use of LIF monoclonal antibodies to block signal transduction pathways in malignant pancreatic cancers, while also assessing their effectiveness on other cancers.
Associate Professor Tian Ruijun’s research team worked with the Salk Institute for Biological Sciences, and specifically the team of Academician Tony Hunter. SUSTech was the second author of the paper. Post-doctoral fellow Gao Weina was the co-second author with Yuan Xiao as the fourth author of the paper. Ph.D. student Huang Peiwu was the fifth author on the paper.
Other universities involved in this paper were the University of California San Diego, the David Geffen School of Medicine at the University of California Los Angeles, Texas Oncology-Baylor University Medical Center, the Eppley Institute for Research in Cancer and Allied Diseases at the University of Nebraska Medical Center, the University of California San Francisco, and the Lunenfeld-Tanenbaum Research Institute, Mount Sinai and Department of Molecular Genetics at the University of Toronto. Additional institutions included Shenzhen People’s Hospital, the Translational Genomics Research Center and HonorHealth.
The work was funded by grants from the Ministry of Science and Technology of China, National Natural Science Foundation of China, NIH grants, the Lustgarten Foundation Award, the Leona M. and Harry B Helmsley Charitable Trust grant, the William Isacoff Research Foundation for Gastrointestinal Cancer grant, Stand Up to Cancer Dream Team Research Grants, the NRSA F31 fellowship award, the Flinn Foundation, the Howard Hughes Medical Institute, the Ipsen/Biomeasure and Freeberg Foundation, the William Isacoff Research Foundation for Gastrointestinal Cancer, the Jeanne Shelby Fund for Cancer Research of Communities Foundation of Texas and the Leona M. and Harry B Helmsley Charitable Trust.
Original article: https://www.nature.com/articles/s41586-019-1130-6
Research Funders: https://standuptocancer.org/press/key-protein-a-possible-new-target-in-the-treatment-of-pancreatic-cancer/
Salk Institute: https://www.salk.edu/news-release/new-study-targets-achilles-heel-of-pancreatic-cancer-with-promising-results/
Fellow researchers: https://azbigmedia.com/tgen-and-honorhealth-contribute-to-nature-study/
American Association for the Advancement of Science: https://www.eurekalert.org/pub_releases/2019-04/si-nst041619.php